 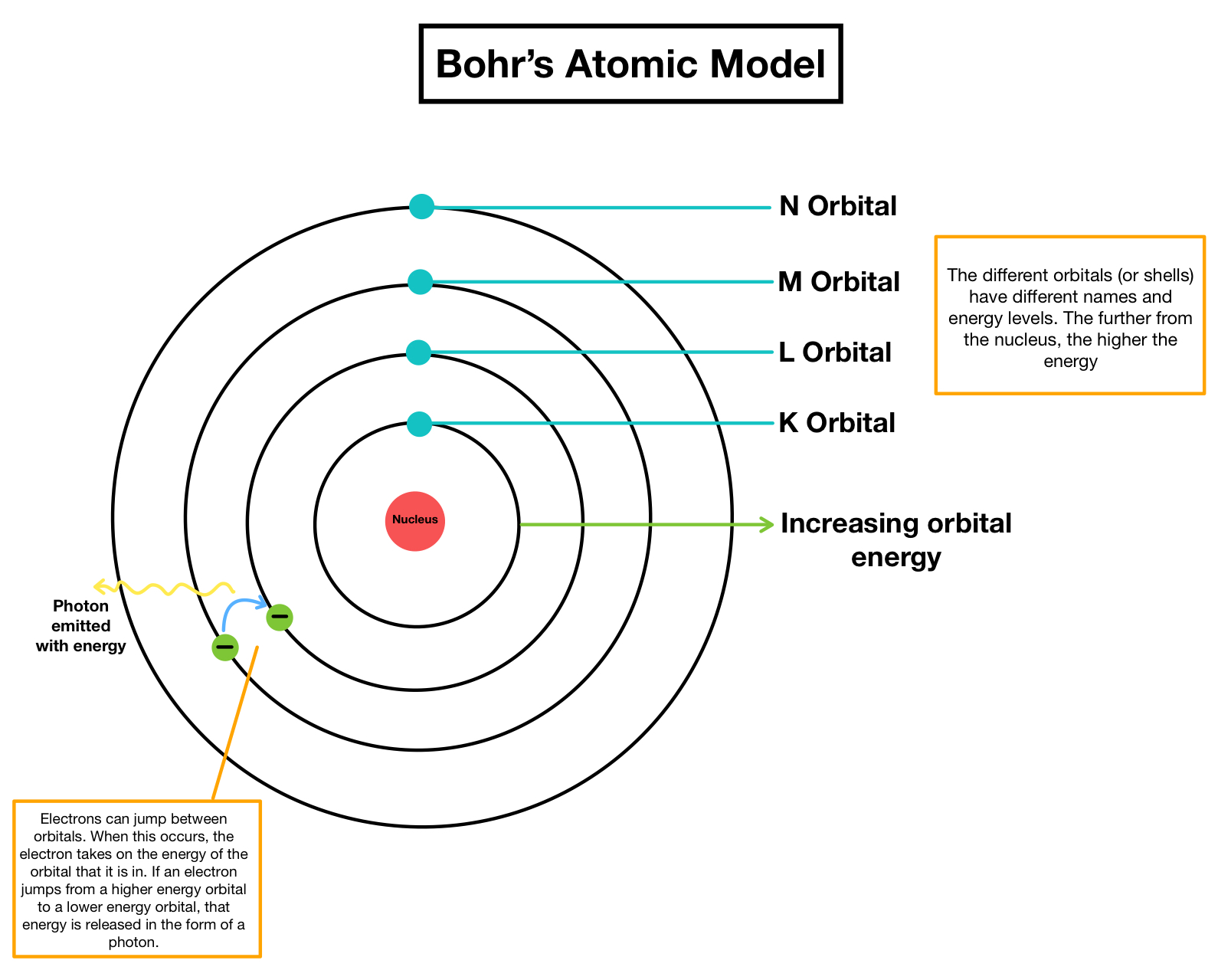 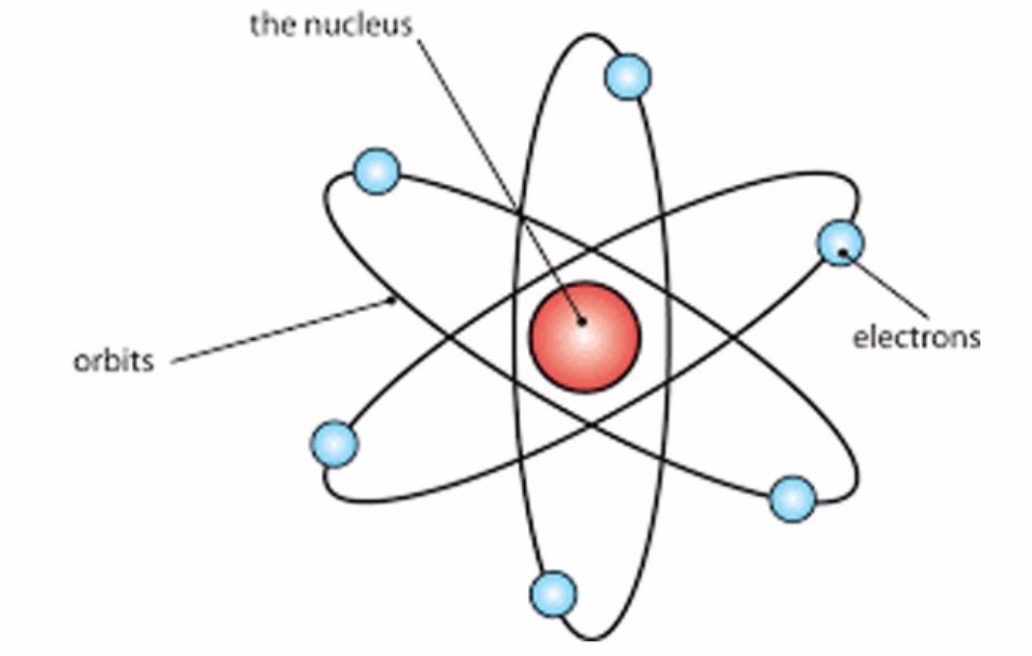
In 1808, John Dalton explained the 3 Fundamental Chemical Laws just discussed, with his Atomic Theory. 
➞ we need to compare the ratio of N atoms by mass in the 3 compounds. Show how this data illustrates the Law of Multiple Proportions. Compound Z consists of 0.4375g N and 1g O. Compound Y consists of 0.8750g N and 1g O. Compound X consists of 1.750g N and 1g O. Unlike the Law of Definite Proportions, here we are considering multiple compounds and the ratio is between two of the same type of element: 2"O" : 1"O".ĭalton began to think that because there was always whole-number multiples, there must be some simple repating unit.įor example, one cannot have CO 1.5 because you cannot have 0.5 of a "unit" (an oxygen atom).Įx: Consider 3 compounds that consist of only N and O atoms. In the multiple compounds above, the ratio of "O" atoms per the one "C" atom is 2:1. Law of Multiple Proportions = when 2 elements form a series of compounds, the ratios of the 2nd elements that combine with the first element can be reduced to small whole numbers. * When John Dalton was on his way to discovering the "atom," he stumbled into, and thus discovered the next law. In other words, the ratio of H:O atoms in water is always 2H : 1O within the one, definite compound, H 2O. Thus, water is always 11.2% H and 88.8% O by mass. ➞ the total is the molar mass, or molecular weight of water = 18.02 g/mol. Law of Definite Proportions = a given compound always contains exactly the same proportion (ratio) of elements by mass. ➞ Since matter can neither be created nor destroyed, we must have four hydrogens and two oxygens on both sides of the equation ~~ on both sides of the chemical reaction. This law is credited to Lavoisier (1743 - 1794).Įx: Consider the decomposition of water into its elements. Law of Conservation of Mass = matter is neither created nor destroyed. In General Chemistry there are 3 Fundamental Chemical Laws to be aware of. Today we are going to discuss the Law of Conservation of Mass, the Law of Definite Proportions, the Law of Multiple Proportions, and Dalton's Atomic Theory. 3 - Chemical Quantities and StoichiometryĤ - Types of Chemical Reactions and Solution Stoichiometryħ - Quantum Mechanical View of the Atom, and Periodicityĩ - Covalent Bonding and Molecular Orbitalsġ0 - Liquids, Solids, and Intermolecular Forcesġ5 - Applications of Acid-Base Equilibriaġ6 - Spontaneity, Entropy, and Free Energyġ8 - Transition Metals and Coordination ChemistryĢ0 - An Introduction to Organic Chemistryġ - Structural, Bonding, Molecular PropertiesĢ - The Nature of Organic Compounds: Alkanes and Cycloalkanesģ - Stereochemistry of Alkanes and Cycloalkanes: 3-D Structures of MoleculesĤ - The Study of Organic Reactions: An Overviewġ0 - Substitution (SN2, SN1) and Elimination (E2, E1) Reactionsġ1 - Mass Spectrometry and IR Spectroscopyġ3 - Conjugated Systems and UV Spectroscopyġ5 - Electrophilic Aromatic Substitution (E.A.S.)ġ8 - Aldehydes and Ketones: Nucleophilic Addition ReactionsĢ0 - Carboxylic Acid Derivatives and Nucleophilic Acyl Substitution ReactionsĢ1 - Carbonyl Alpha-Substitution Reactions 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
